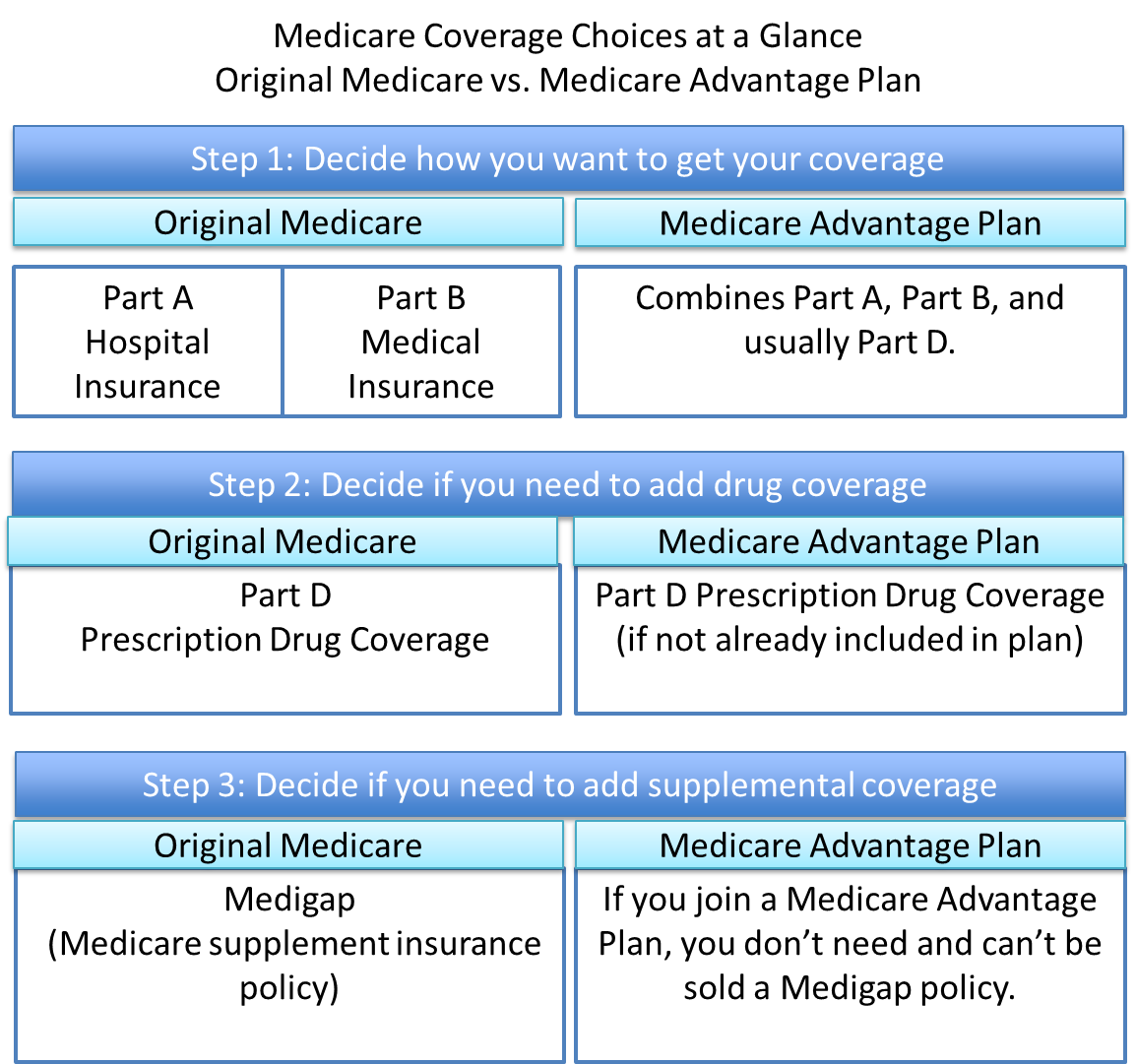
Additional Changes to an Approved Formulary-Biosimilar Biological Product Maintenance Changes and Timing of Substitutions (§§ 423.4, 423.100, and 423.120(e)(2)) Codification of Complaints Resolution Timelines and Other Requirements Related to the Complaints Tracking Module (CTM) ( 42 CFR 417.472(l), 422.125, 423.129, and 460.119) Drug Management Program Notices: Timing and Exceptions § 423.153(f)(8) Definition of Exempted Beneficiary § 423.100 Improvements to Drug Management Programs (§§ 423.100 and 423.153)

National Council for Prescription Drug Programs (NCPDP) Formulary and Benefit Standard, Implementation Guide, Version 60, April 2023 (Approval Date for ANSI: April 12, 2023).National Council for Prescription Drug Programs (NCPDP) Real-Time Prescription Benefit Standard, Implementation Guide, Version 13, July 2023 (Approval Date for ANSI: May 19, 2022).National Council for Prescription Drug Programs (NCPDP) SCRIPT Standard, Implementation Guide, Version 2023011, April 2023 (Approval Date for ANSI: January 17, 2023).
Incorporation by Reference ( 45 CFR 170.299) NCPDP Formulary and Benefit (F&B) Standard Version 60 ( 45 CFR 170.205(u)) NCPDP Real-Time Prescription Benefit (RTPB) Standard Version 13 ( 45 CFR 170.205(c)) Proposal To Adopt Standards for Use by HHS Alignment With Federal Advisory Committee Activities Adoption of Standards and Implementation Specifications Adoption of Health IT Standards and Incorporation by Reference ( 45 CFR 170.205 and 170.299) Incorporation by Reference and Availability of Incorporation by Reference Materials

Summary of Standards for Electronic Prescribing Proposals Date for Required Use of NCPDP SCRIPT Standard Version 2023011, NCPDP RTPB Standard Version 13, and NCPDP F&B Standard Version 60 Requiring NCPDP Formulary and Benefit Standard Version 60 and Retirement of NCPDP Formulary and Benefit Standard Version 3.0 Requiring NCPDP Real-Time Prescription Benefit (RTPB) Standard Version 13 Requiring NCPDP SCRIPT Standard Version 2023011 as the Part D Electronic Prescribing Standard, Retirement of NCPDP SCRIPT Standard Version 2017071, and Related Conforming Changes in § 423.160 Withdrawal of Previous Proposals and Summary of New Proposals Standards for Electronic Prescribing (§ 423.160) Expanding Network Adequacy Requirements for Behavioral Health Enhancements to the Medicare Advantage and Medicare Prescription Drug Benefit Programs Strengthening Current Medicare Advantage and Medicare Prescription Drug Benefit Program Policies: Past Performance Standardize the Medicare Advantage (MA) Risk Adjustment Data Validation Appeals Process Contracting Standards for Dual Eligible Special Needs Plan Look-Alikes For D–SNP PPOs, Limit Out-of-Network Cost Sharing Increasing the Percentage of Dually Eligible Managed Care Enrollees Who Receive Medicare and Medicaid Services From the Same Organization Additional Changes to an Approved Formulary-Substituting Biosimilar Biological Products Enhance Enrollees' Right To Appeal an MA Plan's Decision To Terminate Coverage for Non-Hospital Provider Services Amendments to Part C and Part D Reporting Requirements Annual Health Equity Analysis of Utilization Management Policies and Procedures Enhance Guardrails for Agent and Broker Compensation Mid-Year Enrollee Notification of Available Supplemental Benefits Special Supplemental Benefits for the Chronically Ill (SSBCI) Improving Access to Behavioral Health Care Providers This repetition of headings to form internal navigation links Headings within the legal text of Federal Register documents. This table of contents is a navigational tool, processed from the Provide legal notice to the public or judicial notice to the courts. Rendition of the daily Federal Register on does not Until the ACFR grants it official status, the XML Legal research should verify their results against an official edition of The official SGML-based PDF version on, those relying on it for The material on is accurately displayed, consistent with While every effort has been made to ensure that Regulatory information on with the objective ofĮstablishing the XML-based Federal Register as an ACFR-sanctioned The OFR/GPO partnership is committed to presenting accurate and reliable Register (ACFR) issues a regulation granting it official legal status.įor complete information about, and access to, our official publications Informational resource until the Administrative Committee of the Federal This prototype edition of theĭaily Federal Register on will remain an unofficial Each document posted on the site includes a link to theĬorresponding official PDF file on. The documents posted on this site are XML renditions of published Federal Register, and does not replace the official print version or the official It is not an official legal edition of the Federal This site displays a prototype of a “Web 2.0” version of the dailyįederal Register.
